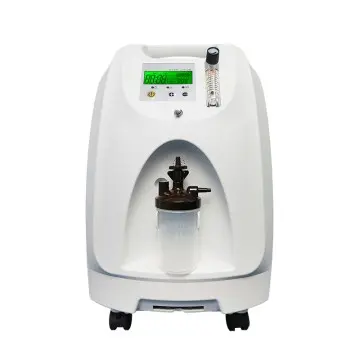
High Flow Portable Oxygen Concentrators from Leading Chinese Factories & Suppliers
The High Flow Portable Oxygen Concentrator from Jiangsu Konsung Bio-Medical Science & Technology Co., Ltd. is an advanced medical device that provides continuous oxygen flow to patients with respiratory conditions. Its lightweight and portable design allow for easy transport, making it an excellent option for those needing oxygen therapy while on the go. Featuring cutting-edge technology, this concentrator delivers a higher oxygen flow than traditional models, ensuring patients receive adequate oxygen for improved lung function and overall health. The user-friendly interface makes operation simple, providing a seamless experience. Suitable for various settings, including home use, travel, and outdoor activities, this concentrator enables patients to maintain active lifestyles while effectively managing respiratory health. With its compact design and extended battery life, the High Flow Portable Oxygen Concentrator ensures convenience and reliability for those in need of oxygen therapy.Step-by-Step Guide To High Flow Portable Oxygen Concentrator From Concept to Delivery Supplier
When it comes to portable oxygen concentrators (POCs), understanding the journey from concept to delivery is crucial for global buyers seeking reliable solutions for home healthcare. The development of a high flow POC involves multiple stages, beginning with thorough market research and identification of user needs. This initial stage is where innovative features such as compact design, ease of use, and high oxygen output are conceptualized. Collaboration with healthcare professionals at this point can guide the creation of a product that truly meets patient requirements.
Following the concept phase, the focus shifts to rigorous R&D and prototyping. This stage emphasizes the importance of testing materials and technologies to ensure safety and efficiency. Involving healthcare providers for feedback during the prototype phase is essential to refine the product further. Once the design is finalized, manufacturing processes must comply with industry standards and regulations, ensuring the highest quality before reaching the market.
Finally, the delivery phase entails a robust supply chain management strategy. Reliable logistics and distribution channels are vital to ensure that high flow portable oxygen concentrators reach healthcare facilities and consumers in a timely manner. Establishing a strong partnership with suppliers and distributors worldwide can enhance market reach and ensure customer satisfaction. This comprehensive approach from concept to delivery not only ensures a successful product launch but also contributes to a seamless user experience in the growing field of home healthcare.
Step-by-Step Guide To High Flow Portable Oxygen Concentrator From Concept to Delivery Supplier
| Phase | Description | Duration | Key Deliverables |
|---|---|---|---|
| 1. Research & Development | Conduct market analysis and define product specifications. | 3 months | Product requirement document |
| 2. Prototyping | Create initial prototypes for testing and feedback. | 2 months | Prototypes and test data |
| 3. Testing & Validation | Perform safety and efficiency tests on prototypes. | 1 month | Test report |
| 4. Manufacturing Setup | Set up production line and finalize supplier agreements. | 4 months | Manufacturing plan |
| 5. Production | Start mass production of the high flow portable oxygen concentrator. | 6 months | Initial batch of products |
| 6. Quality Assurance | Implement quality control measures and final inspections. | 2 months | Quality assurance report |
| 7. Delivery & Logistics | Organize logistics for product delivery to suppliers and distributors. | 1 month | Distribution plan |
| 8. Post-Launch Support | Provide customer support and gather user feedback. | Ongoing | User feedback report |
Exploring High Flow Portable Oxygen Concentrator Now Trending Factory
M
Maria Rodriguez
I love the product! The customer service was exceptional, with knowledgeable staff guiding me through.
22
May
2025
H
Hannah Rodriguez
Very impressed with both the product and customer service. They are knowledgeable and responsive.
14
June
2025
A
Ava Martinez
Fantastic service and superb product quality! Definitely a brand I will trust!
09
June
2025
B
Benjamin Nelson
Excellent quality! I was very pleased with the way the service team resolved my issues.
02
July
2025
L
Lily Walker
This product is a standout! The after-sales team provided prompt and professional assistance.
14
May
2025
J
Joshua Scott
Superior quality! The after-sales support team were attentive and professional.
19
June
2025